NABP Pilot Brings Trading Partners, Regulators Together for Public Health Protection
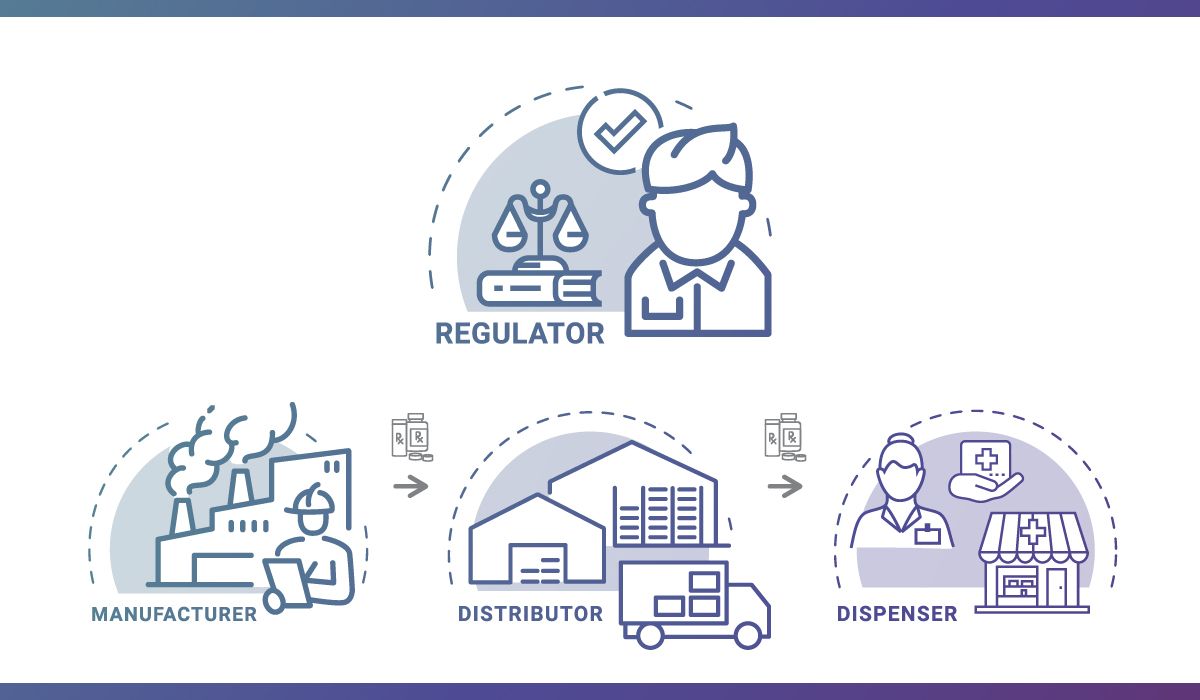
By November 27, 2023, the Food and Drug Administration’s (FDA) Drug Supply Chain Security Act (DSCSA) will be fully implemented. We’ve been working with the boards of pharmacy, other regulators, and trading partners to prepare for the requirements included in Title II of the Act, which calls for product tracing at the package level. Our […]
Read More ›New Business Models, Revised Regulations Improving Pharmacy Access in Rural Communities

For many people living in rural areas, access to pharmacy services is difficult. The University of Iowa’s Rural Policy Research Institute Center for Rural Health Policy Analysis reports that more than 16% of the independently owned rural pharmacies in the US closed between March 2003 and March 2018, lowering the number to fewer than 6,400. […]
Read More ›Combatting Opioid Epidemic Still Top Priority for Ohio Board

Boards of pharmacy can face unexpected challenges while working to protect the public health. Here, board executive directors talk about how they worked around these obstacles. Steven W. Schierholt, Esq Executive Director, State of Ohio Board of Pharmacy How long have you served as executive director of the State of Ohio Board of Pharmacy? What […]
Read More ›Newsletter Refresh Keeps Licensees Engaged with State Updates
In July this year, our state newsletters debuted an eye-catching, easy-to-follow layout. It was their first redesign since 2010. Our state newsletter program helps boards of pharmacy disseminate regulatory and practice updates vital to each state’s pharmacists, pharmacy technicians, and other licensees. The 33 participating boards provide us with their news and information and we compile […]
Read More ›How the California Board Uses Waivers to Help Patients During COVID-19

Boards of pharmacy can face unexpected challenges while working to protect the public health. Here, board executive directors talk about how they worked around these obstacles. Anne Sodergren Executive Officer, California State Board of Pharmacy How long have you served as executive officer? I was appointed to serve as executive officer in January 2020. Prior […]
Read More ›Truths and Misconceptions About FDA’s Compounding MOU
Earlier this month, FDA announced it will provide states one more year to decide whether to sign its compounding memorandum of understanding (MOU) before the agency begins enforcing the statutory 5% limit on out-of-state distribution of compounded human drug products. This comes after many states asked for more time to wrangle with the legal and […]
Read More ›We Offer Helping Hand to States Building Drug Importation Plans
Prescription drug prices continue soaring and no light is visible at the end of the tunnel. Wanting to bring relief to cash-strapped constituents, many states are creating their own drug importation programs from Canada using an FDA rule finalized in November 2020. Regulating these systems will be complex, but states do not have to go it alone. That’s why I’m focusing my term as NABP President on helping them navigate the process to ensure the integrity of the drug […]
Read More ›To Protect Public Health, NABP Clearinghouse Helps Boards Track Administrative and Disciplinary Data

One of the most important responsibilities a board of pharmacy has is ensuring its thousands of pharmacists, pharmacy interns, pharmacy technicians, facilities, and more practice safely and legally. This is a daunting task within itself, but we have a tool that can help. NABP Clearinghouse eases the workload. It’s a national database of disciplinary information […]
Read More ›Keeping Pharmacies Safe in Kentucky

Inspectors/investigators are the eyes and ears for boards of pharmacy, ensuring compliance with pharmacy law and practice standards. In this feature, inspectors reveal what they’ve learned in the field. Katie Busroe, RPh, BCSCP Inspections and Investigations Supervisor, Kentucky Board of Pharmacy How long have you been serving as an inspector for the Board? What was […]
Read More ›New Department Has Member Boards Covered
Out of the many things that the COVID-19 pandemic has taught us, one is that no individual or group is an island. And this adage was reinforced for our member boards of pharmacy when the pandemic first tightened its grip in March 2020. Many contacted us for tips on handling stay-at-home orders, COVID-19 testing and […]
Read More ›